Atrial Fibrillation (A-Fib) is sustained cardiac arrhythmia, affecting 6% of the population over the age of 65. It is the leading cardiac cause of stroke and increases the risk of stroke five-fold. It is also associated with heart failure and consequently is a considerable burden on health service resources.
The ventricular rate can be controlled either by using antiarrhythmic drugs or by catheter ablation of the atrioventricular (AV) node and pacemaker implantation.
However, this does not restore atrial transport nor does it eliminate the risk of embolic strokes, even with the use of anticoagulants. Therefore, restoring and maintaining Sinus Rhythm (SR) is the optimal treatment.
One of the most exciting and promising treatments for the cure of A-Fib is the use of catheters to apply radiofrequency energy to the atrial tissue. This modifies the electrical proper ties of the atrium in order to eliminate the tendency to fibrillate.
There is increasing confusion regarding the results and benefits of catheters for A-Fib because of the rapidly evolving techniques, and a wide range of opinion and recommendations regarding its efficacy. The number of catheter ablation procedures is increasing dramatically. An apparently chaotic heart rhythm is amenable to cure, and in order to understand how, the currently employed catheter techniques must be reviewed.
See Also:
PAROXYSMAL, PERSISTENT AND PERMANENT
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataA-Fib is usually prefixed by a temporal descriptive term. Paroxysmal A-Fib terminates spontaneously. Persistent A-Fib describes A-Fib lasting for more than seven days or requiring some intervention to terminate it.
Permanent A-Fib is resistant to all conventional interventions to terminate it. These conventional interventions include anti-arrhythmic drug therapy and direct current cardioversion.
UNDERLYING MECHANISMS
The mechanism of A-Fib is not clearly understood. The most commonly accepted mechanism underlying A-Fib is multiple, random waves of electrical depolarisation.
These waves follow re-entry circuits around lines of conduction block in the atria, which may be fixed (present all the time, like anatomical structures such as veins) or functional (only present under certain circumstances, depending on the conduction property of the myocardium).
It has become apparent, however, that the initiating event is the presence of rapidly firing premature atrial beats, which most often arise from atrial tissue within the pulmonary veins, the veins that drain blood from the lungs into the left atrium. These are called focal ectopic tachycardias.
In some patients it has also been observed that this rapidly firing focal activity can also be responsible for maintaining A-Fib as well as initiating it. This focal activity gives cardiologists a potential target for curative catheter ablation for the first time.
CATHETER ABLATION OF A-FIB
Curative ablation of A-Fib has evolved to encompass two goals: removing all potential triggers that may initiate or perpetuate A-Fib and altering the conduction properties of the atria (substrate modification) so that AFib cannot be sustained even when triggered.
It has become apparent that patients have a wide range of mechanisms for their atrial fibrillation and the clinical presentation is a crude and perfect guide to the possible mechanism.
For example, those patients presenting with frequent non-sustained episodes of A-Fib are more likely to have a repetitive trigger leading to their problems.
In contrast, the abnormal electrophysiological properties of the atrium are likely to be more important in patients with permanent A-Fib. The recognition of the range of mechanisms depending on type of A-Fib has allowed procedures to be tailored so that, for example, in paroxysmal A-Fib only an isolation of triggers is attempted and not the more complex substrate modification.
ABOLITION OF TRIGGERS
The most common sources of triggers are the pulmonary veins (PVs), but up to 20% may be non-PV in origin, such as the superior vena cava, coronary sinus or crista terminalis. These triggers are rarely active during a catheter ablation procedure, so the approach to catheter ablation is to treat the most common aetiology, the PVs.
The trigger sites are isolated from the rest of the atria either by destroying the muscular connections that link them to the atria or creating a continuous line of conduction block in the atria that surrounds and encloses them.
This is done in two ways: either by delivering energy close to the vein origin or creating a wide circle of energy application which encloses ipsilateral vein origins. Although there remains controversy as to whether true electrical disconnection is necessary to achieve success, most key opinion leaders believe that it is.
In order to confirm electrical isolation, a circular mapping catheter is advanced to the pulmonary veins and used to record electrical activity. When the electrograms on the mapping catheter disappear, the vein is isolated.
Using ablation to abolish triggers produces excellent results (51%–100% freedom from A-Fib). However the methods and reporting of observational studies vary greatly.
SUBSTRATE MODIFICATION
There are two approaches to substrate modification. Firstly it can be achieved by creating transmural linear lesions that connect two anatomical structures and form barriers to conduction, thus interrupting the re-entry circuits that perpetuate A-Fib.
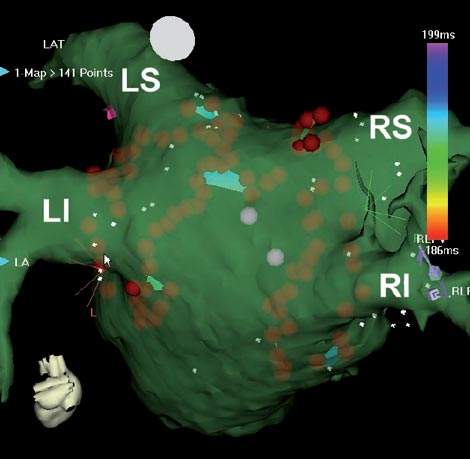
The alternative approach (also known as atrial debulking) is to reduce the amount of atrial tissue available to form re-entry wavefronts, either by enclosing and isolating large areas of atrial tissue (typically done around the PVs) or ablating widely all over the atria, thus reducing the amount of viable tissue. This is usually done with the assistance of a non-fluoroscopic mapping system.
These systems allow three dimensional imaging of the ablation catheter without the need for x-rays and allow accurate placement of lesions within the atria, but also provide a record of where lesions have already been placed. A possible downside to substrate modification is the reduction in the contractile potential of the atria, which may prevent recovery of mechanical function.
It is clear from observational studies that PV isolation alone is not as effective for patients with persistent or permanent A-Fib in comparison with paroxysmal A-Fib, which is consistent with the current understanding of the mechanisms described above. Initial attempts at delivering long lines of radiofrequency ablation to connect anatomical obstacles, although successful, led to high complication rates.
Wide encirclement success has been reported as high as 68% freedom from permanent A-Fib and 73% from non-paroxysmal A-Fib. With the addition of proven electrical isolation, even permanent A-Fib can be successfully abolished in 79% of patients after multiple procedures.
However, achieving this is technically challenging and requires long, arduous procedures. In addition, gaps in these lines may promote regular atrial tachycardias, which use the ablation lines as their pathway.
An alternative approach has been the ablation of all fragmented electrical activity in the right and left atrium, with the hypothesis being that these are consistent sites where fibrillating wavefronts turn or split. By ablating these areas, the propagating random wavefronts are progressively restricted until the atria can no longer support A-Fib.
It is not clear whether the success of this technique is related simply to debulking myocardium or to targeting the critically positioned electrograms.
NEW TECHNOLOGIES
A number of technological advances have contributed to the increased success of A-Fib ablation, but probably the most significant are advancements in three-dimensional catheter imaging technologies. It is now possible to import and register a three-dimensional CT or MRI scan into a catheter imaging system so that the catheter can be manipulated in a three-dimensional rendering of the patients left atrium.
Other technologies, such as the use of ultrasound balloons to isolate the veins, show promise but are still under development and unproven.







