The landscape of medical imaging has changed dramatically from the first plain film radiographs to today’s advanced imaging systems, which include ultrasound, MRI, multidetector CT and nuclear, and molecular imaging devices such as PET and PET/CT.
As these changes have evolved, so have the decision algorithms for choice of modality and technique of scanning to better serve the intended recipient: the patient. As the newest technology, PET/CT has rapidly progressed within a short period, popular with imagers, primary clinicians and researchers. However, there are questions of efficacy in the face of methods and modalities in practice, and as the best ways to use the technology in the modern medical setting.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
PET background
The origin of PET imaging began shortly after that of CT in the early 1970s. By the early 1980s, PET was showing promise in the arena of neurological imaging of seizure disorder as well as aiding in evaluation of malignancies of the brain. The late 1980s saw the advancement of whole body PET imaging and soon PET was used for the detection and evaluation of many oncologic and non-oncological disorders. However, technology was adopted by the medical community at a slower rate than its radiological counterparts CT and MRI.
Barriers to more widespread usage included equipment and facility costs, adequate patient referral concerns, radiopharmaceutical supply issues, interpreter training issues and image quality. These issues served to restrict the deployment of PET to larger research centres, academic institutions and some private imaging centres, and moved the modality into a more ancillary study type rather than a mainstream modality.
Like many nuclear medicine modalities, detailed anatomic structure resolution is lower with PET when compared with CT or MRI simply by the nature of the technologies involved. However, the power provided by functional nuclear medicine scans, including PET, often trumps the static anatomic resolution of the aforementioned modalities. The integration of the anatomic and the physiologic sides of the imaging picture became an important chasm to cross in order to increase the penetration of PET
into mainstream medicine.
Coregistration and fusion of mixed modality datasets came into practice in the 1990s with impressive results, but the computing processing power required, as well as the time cost to perform these functions, was often prohibitive. There were also difficulties in proper coregistration for non-static areas of the chest, abdomen and pelvis where patient motion, normal physiologic functions and positioning would reduce the accuracy of the coregistration and fusion process.
In 1998, the first hardware-driven mixed modality PET/CT scanner was installed at the University of Pittsburgh, Philadelphia, and evaluating clinical patients began in May that year. It was lauded as a powerful tool to accurately view a coregistered high quality anatomic and functional combined scan. In addition, the ability to leverage the CT scan as an effective and rapid tool for attenuation correction, improved PET image quality and speeded patient throughput. Even in this early phase of
PET/CT clinical evaluation, the initial patient group studied had an impact in patient clinical management of 30%.
PET/CT issues
As the results of this pilot device reached the attention of imaging centres, feedback came in the form of purchase orders for some and cautious reserve for others. The potential applications in the clinical arena were impressive both in and out of the scope of oncology, however, the adoption of the new device design was not without its issues.
Firstly, the cost of installing a dedicated PET scanner with support of either an on-site radiopharmacy or regional distribution chain was high on its own. The addition of the CT portion greatly increased the unit cost as well as the size, weight and shielding requirements to install this hybrid scanner. Changes in the technical type of CT attached, in terms of number of detectors, speed and number of sources, complicates the purchasing decision depending on whether the scanner is to be used
solely for PET/CT purposes, mostly CT with some PET/CT or PET/CT with some CT.
PET/CT with some CT is the dominant usage model. As well as the design issues with CT, the PET portion has also seen changes in design. Different detector crystals, reconstruction algorithms, electronic packages and gating devices that were changing on an annual basis served as an indication for many institutions to hold off purchases until pricing and design stabilised.
Although the variety of designs still persists, the options have lessened as designs have converged, making purchasing decisions easier. Costs have also stabilised for scanners with cutting-edge models in the range of $2m and older models (or ones with smaller CT packages) dropping below that.
While the scanner equipment pricing has stabilised to a degree, there has been a reduction in the costs of fluorodeoxyglucose (FDG) and improved availability. The PET radiotracer, FDG, is an injectable glucose analogue. PET scanners can image FDG to evaluate relative glucose metabolism. Other radiotracers allow imaging of different physiology.
This applies to on-site cyclotron purchases as well as to a much larger degree for regional distribution centres. The drop in FDG costs can largely be attributed (at least in the US) to a boom in recent PET/CT deployment, thus a volume purchasing effect. This increase in adoption in the US has also changed the sales data for the type of PET-based device. In 2003, 79% of all PET scanners purchased were of the PET/CT variety and in 2007 this number has grown to over 99%, and most manufacturers
have stopped production of clinical dedicated PET-only scanners.
There has also been an increase in overall purchases and use with growth rates well over 10% per year worldwide. The increase in demand from the medical community has caused some problems with deployment. As more scanners find their way into community hospitals, commercial imaging centres are seeing increased competition for patient referrals. The push for new deployment of scanners can cause geographical variations with high densities of scanners in some areas and scarcity in others.
As with any substantial capital investment, the decision to purchase and install a PET/CT scanner in the hospital or non-hospital setting should be carefully planned. Outside the logistical considerations of PET/CT installation the root of interest in the technology must and does come from the ability of the modality to positively affect the patient and overall costs of the patient’s care.
Installation growth
Despite the logistical and financial issues involved with the deployment of PET/CT scanners, there has been a dramatic upswing in the number of installed scanners largely due to what PET/CT adds to patient care versus dedicated
PET imaging alone or anatomic imaging alone. Many arguments have been voiced over the issues of PET/CT with respect to technique of including (or not) IV contrast with the CT scan portions, the inclusion of standardised uptake value measurements and scan times for individual bed positions. However, these technical issues do not detract from PET/CT’s ability to answer difficult questions that other modalities cannot do with as much certainty (Figure 1).
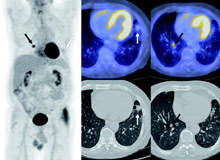
In diagnosis, PET/CT can classify an abnormality as benign versus malignant, infectious versus non-infectious, inflammatory versus non-inflammatory, or metabolically active or not. In some cases, PET/CT can have false positive results in distinguishing inflammation versus malignancy, particularly in the thorax. PET/CT can also help diagnose conditions that would not meet criteria by size characteristics on CT in the setting of significant hypermetabolic activity (Figure 2).
When evaluating treatment efficacy for oncology, the combination of both physiologic data and anatomic resolution that FDG PET/CT offers, trumps most single modality scans. The ability to track lesions over time and to track both metabolic changes (in FDG) and size changes allows treatment management changes to be made earlier. Detecting early decreases in metabolic rates can separate patients with a good therapeutic response from those that might require a change in treatment regimens,
resulting in prolonged survival (Figure 3).
PET/CT is an effective tool in surveillance for post-therapy patients at regular intervals, giving the patient a quick, single encounter to complete their scanning needs. When all these facets of FDG PET/CT are added up, the benefit of the PET/CT scan is clear. This does not imply that PET/CT should supplant standard CT or MRI, as the CT portion of PET/CT is optimised for the PET technique. However, it can cut down the need for additional CT scans when a PET/CT is present.
Patient care savings
On a per-scan basis, PET/CT is expensive. However, when factoring the positive impact that PET/CT scans can have on the overall management of a patient, chemotherapy and radiation therapy, the end line cost savings for the patient’s total care can be substantial.
For example, in a study of B-cell lymphoma patients, the expected cost savings for the studied patient population was $156.2m, or $8,239 per patient, due to better identification of chemo-refractive patients by PET/CT from the CT scan alone.
The overall cost savings extend beyond the patients actively receiving care to include those who would need care in the future. This is achieved by channeling patients into the most appropriate therapies and resources, and away from treatments that would not be as effective, thus freeing resources for other patients.
Conclusions
PET/CT is a powerful tool used in initial staging, re-staging, therapy efficacy evaluation and post-therapy surveillance for most oncological disorders. FDG PET/CT is here to stay. Its use will grow as the positive impact it can have on patients outweighs any of its shortcomings or financial/logistical hurdles and has a solid position to work in tandem with existing CT and MRI modalities.
Research material has been referenced in this text. For full details please contact the editor, andrewtunnicliffe@spgmedia.com





