Antimicrobial agents, which include antibiotics and similar drugs, have been used for the last 70 years to treat infectious diseases. The drugs have been used so widely and for so long that the germs they are designed to kill have adapted to them, making the drugs less effective. In 2013, the Centers for Disease Control and Prevention (CDC) identified 18 drug-resistant threats in the US and categorised each one based on level of concern: urgent, serious, and concerning. Threats labelled as urgent are high-consequence antibiotic-resistant threats and have significant risks across several criteria. Among the list of urgent threats is carbapenem-resistant Enterobacteriaceae (CRE).
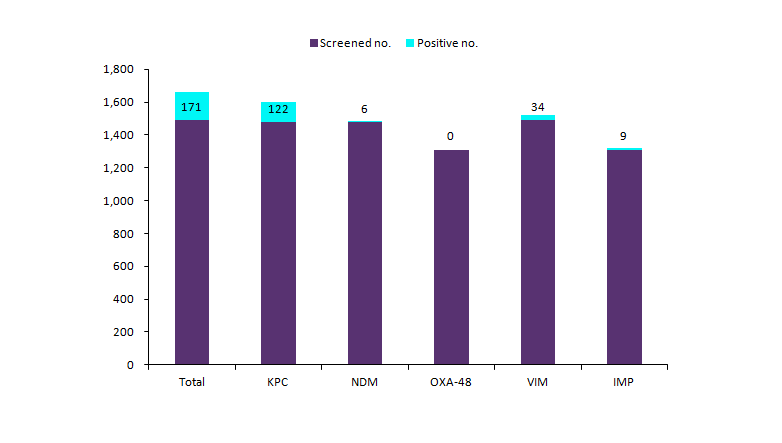
A recently published Morbidity and Mortality Weekly Report (MMWR) evaluated infection data from the National Healthcare Safety Network from 2006 to 2015 to analyse changes in the annual proportion of selected pathogens that were nonsusceptible to extended-spectrum cephalosporins (ESBL phenotype) or resistant to carbapenems (carbapenem-resistant Enterobacteriaceae). The study found that the percentage of carbapenemase-producing isolates was highest among Klebsiella species. Among carbapenemase-producing carbapenem-resistant Enterobacteriaceae, the most commonly identified carbapenemase was Klebsiella pneumoniae carbapenemase (see Figure 1). Klebsiella, a common Gram-negative bacteria, is becoming increasing difficult to treat. It is increasingly becoming resistant to antibiotics, including carbapenems. Patients with antibiotic-resistant Klebsiella are over twice as likely to die from the infection compared to those with non-resistant Klebsiella. The CDC reports that CRE bacteria is on the rise among patients in medical facilities and almost half of hospital patients who get bloodstream infections from CRE bacteria die from the infection. This is alarming because carbapenem antibiotics are usually the last line of defense against Gram-negative infections that are resistant to other antibiotics.
Figure 1: Screening Tests for Carbapenem-Resistant Enterobacteriaceae Colonization, Antibiotic Resistance Laboratory Network Laboratories and CDC Laboratory, Specimens Collected January 1–September 30, 2017

| Source: GlobalData; CDC. |
Abbreviations: IMP = imipenemase ; KPC = Klebsiella pneumoniae carbapenemase; NDM = New Delhi metallo-beta-lactamase; OXA-48 = oxacillinase-48-like carbapenemase; VIM = Verona integrin encoded metallo-beta-lactamase
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataThe MMWR found that post-acute care facilities had the highest percentage of carbapenem-resistant Enterobacteriaceae, compared to short-term acute care hospitals. There are more challenges in post-acute care facilities that may amplify transmission of the resistant organisms, such as a longer duration of stay, prioritising resident quality of life and therefore less aggressive use of transmission-based precautions, high staff turnover rates, and less expertise and training in infection controls. It is recommended that healthcare facilities address transmission problems in a timely and comprehensive manor.
Each year in the US, at least two million people become infected with antibiotic-resistant bacteria and at least 23,000 people die each year as a result of the infections. However, the US is not alone—antibiotic resistance is a global threat. The misuse of antibiotics in humans and animals may be accelerating the process of antibiotic resistance. The CDC estimates that up to 50% of the time that antibiotics are prescribed, they are not needed and are incorrect when it comes to dosing or duration. The misuse and overuse of antibiotics leads to higher medical costs, longer hospital stays, and increased mortality.
The CDC is working with other countries to strengthen international collaboration and capacities for antibiotic resistance prevention, surveillance, control, and R&D. The World Health Organization (WHO) has also been leading a number of initiatives to address antimicrobial resistance. In order to prevent the spread of antibiotic resistance, there are prevention and control measures that can be taken. Individuals should only use antibiotics when prescribed by a certified health professional. Policy makers can improve the surveillance of antibiotic-resistant infections and strengthen policies. Health professionals should only prescribe antibiotics when they are needed, according to current guidelines, and talk to their patients about how to take them correctly. The healthcare industry can invest in R&D of new tools and new medicines. The agricultural sector should properly use antibiotics under veterinary supervision and vaccinate their animals to reduce the need for antibiotics. As antibiotic/antimicrobial resistance continues to grow as a global problem, it is important to address current problems before it is too late.
For more insight and data, visit the GlobalData Report Store – Verdict Hospital is part of GlobalData Plc.