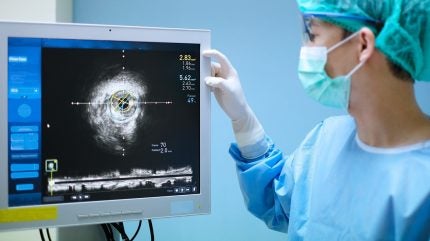
Abbott has received the US Food and Drug Administration (FDA) approval and CE Mark for its Ultreon 3.0 AI-powered coronary imaging platform.
The approval enables the integration of automated imaging insights with real-time planning guidance during percutaneous coronary intervention (PCI) procedures to support clinicians and potentially improve patient outcomes.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The next-generation Ultreon 3.0 uses AI with optical coherence tomography (OCT) to deliver high-resolution, real-time imaging during minimally invasive procedures that address blocked heart arteries.
The system is designed to support a fully integrated clinical workflow to enable faster, precise coronary interventions.
One feature of the Ultreon 3.0 software is its one-second OCT pullback, a high-speed, infrared light-based imaging technique.
This captures detailed cross-sectional views of coronary arteries while offering higher resolution than intravascular ultrasound (IVUS) and the capability for low or zero contrast use.
Reducing the amount of contrast may be especially useful for patients with kidney disease, who comprise around 25% of those with coronary artery disease.
Key capabilities of Ultreon 3.0 include detailed assessments of blockage size, shape, and location to support precise stent deployment, as well as post-procedure assessments to confirm improved blood flow.
The system features a streamlined set-up, advanced AI-driven workflow efficiencies, and the ability to efficiently plan procedures for complex coronary cases.
Abbott vascular business chief medical officer Ethan Korngold said: “Complemented by our leading coronary portfolio, Ultreon 3.0 makes OCT imaging more intuitive and powerful for physicians, reimagining imaging into a tool for precision-guided intervention before and after procedures.
“Advancements like Ultreon 3.0 showcase Abbott’s leadership in AI-powered imaging and as a trusted partner in delivering smarter, more personalised coronary care.”
Ultreon 3.0 builds on the previous version by introducing enhanced AI capabilities targeted at the needs of complex coronary PCIs.
Last month, Abbott announced the integration of its Precision Oncology portfolio into Flatiron Health’s cloud-based electronic medical record (EMR) platform, OncoEMR.



