GE HealthCare has dosed the first patient in its international, multi-centre Phase II/III LUMINA clinical trial for the investigational manganese-based magnetic resonance imaging (MRI) contrast agent, mangaciclanol.
The trial commenced at Mayo Clinic in Rochester, Minnesota, US, targeting adults and paediatric patients aged two years and older, following fast-track designation by the US Food and Drug Administration (FDA).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
If approved, mangaciclanol may serve as an alternative to gadolinium-based MRI contrast agents currently used worldwide. The FDA fast-track status aims to speed up the evaluation of therapeutics that address serious unmet needs.
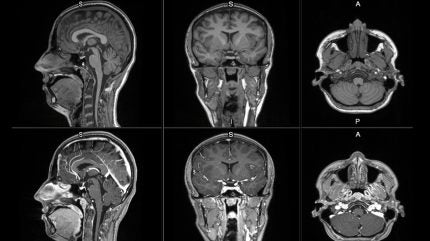
The new contrast agent is intended to improve MRI detection and visualisation of lesions with abnormal vascularity in the central nervous system and body.
MRI contrast agents help enhance the visibility of abnormal structures during scans, supporting improved differentiation between healthy and unhealthy tissue.
Early clinical images of mangaciclanol indicate comparable relaxivity to gadobutrol, a widely used gadolinium-based agent, with similar diagnostic potential.
Unlike gadolinium, which is a rare-earth metal, manganese is an essential element in food, endogenously regulated in the human body, and the macrocyclic structure of mangaciclanol is designed to limit retention.
Contrast agents are necessary in nearly one-third of MRI procedures globally, with an estimated 65 million gadolinium-enhanced studies carried out each year.
Gadolinium supplies rely heavily on mining operations in China, presenting a potential supply risk. Manganese, however, is more widely available from sources in Australia, Gabon, and South Africa, helping to mitigate such risks.
The agent could also address environmental concerns, as manganese is naturally present in water sources, potentially lessening the impact of excreted contrast media on groundwater.
GE HealthCare president and CEO Peter Arduini said: “As demand for diagnostic imaging continues to rise, we continue to advance our imaging agent pipeline to better meet the needs of patients.
“This clinical milestone builds on GE HealthCare’s leadership in contrast media, as mangaciclanol has the potential to transform the MR imaging market and strengthen the resiliency of its supply chain.”
Mangaciclanol’s Phase I trial results showed that the agent was well tolerated, with no serious adverse events, dose-limiting toxicities, or clinically significant findings observed.
GE HealthCare’s Pharmaceutical Diagnostics unit is a recognised provider of imaging agents, supporting 140 million patient procedures annually.
For more than 40 years, GE HealthCare contrast media has been used in MRI, ultrasound, and X-ray/CT imaging.
Mangaciclanol remains in clinical development and is not yet approved for commercial use.
Last week, GE HealthCare expanded its partnership with RadNet subsidiary DeepHealth to broaden access to AI-powered breast cancer screening solutions.



