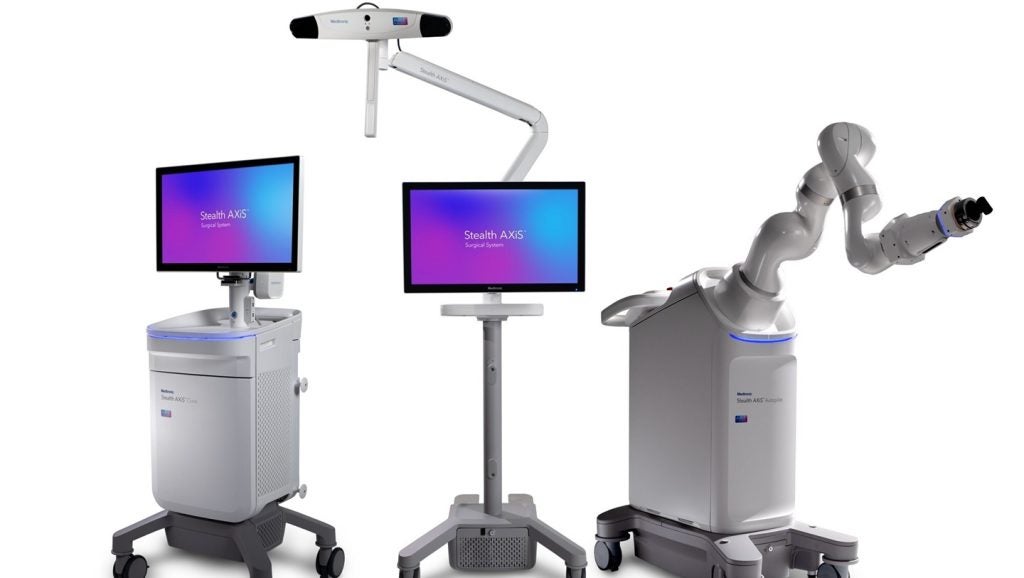
Medtronic has received the US Food and Drug Administration (FDA) clearance for the Stealth AXiS spine surgery robotics platform.
The system is designed as an integrated solution that combines robotics, planning, and navigation in a single platform for spine procedures in the US.
Medtronic aims to enable hospitals and ambulatory surgery centres to perform these procedures without relying on multiple separate technologies.
The system’s architecture allows support for a variety of surgeon preferences and clinical complexities.
Medtronic has built this platform to meet current needs while offering the possibility of future expansion to cranial and ear, nose, and throat (ENT) applications, subject to further 510(k) clearance.
The modular design allows institutions to deploy only what they need now and scale capabilities as their clinical requirements change.
A significant feature of the Stealth AXiS system is LiveAlign segmental tracking, which provides real-time visualisation of anatomical movement, surgical adjustments, and patient alignment during spine surgery.
This functionality reduces the need for repeated imaging and minimises reliance on manual workflow steps, supporting consistent execution of patient-specific surgical plans.
The Stealth AXiS system forms a core part of Medtronic’s AiBLE smart ecosystem by integrating planning, navigation, and execution into a single workflow. This approach streamlines surgical processes and enables seamless information sharing before, during, and after surgery.
The native connectivity within the AiBLE ecosystem is designed to link devices, software, and data to facilitate more meaningful insight exchange across the surgical continuum.
Medtronic senior vice-president and cranial and spinal technologies president Michael Carter said: "Spine surgery is complex, and variability remains a real challenge.
“The Stealth AXiS system is designed to make advanced technology more usable and clinically meaningful, helping surgeons deliver more predictable, personalised care while laying the foundation for continued innovation."
Earlier this month, Medtronic reported three milestones in the US, expanding access to its MiniMed 780G insulin delivery system for individuals with type 1 diabetes (T1D) and insulin-requiring type 2 diabetes (T2D).